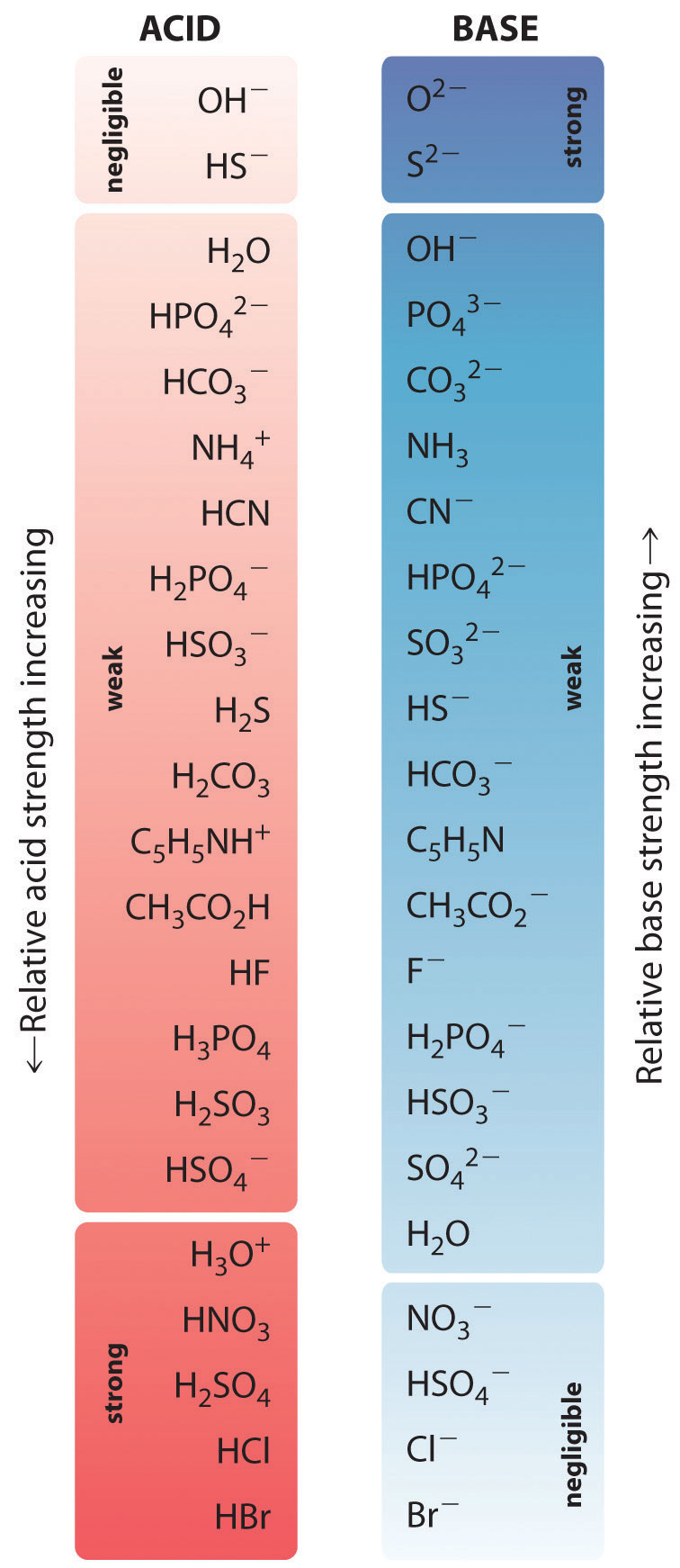
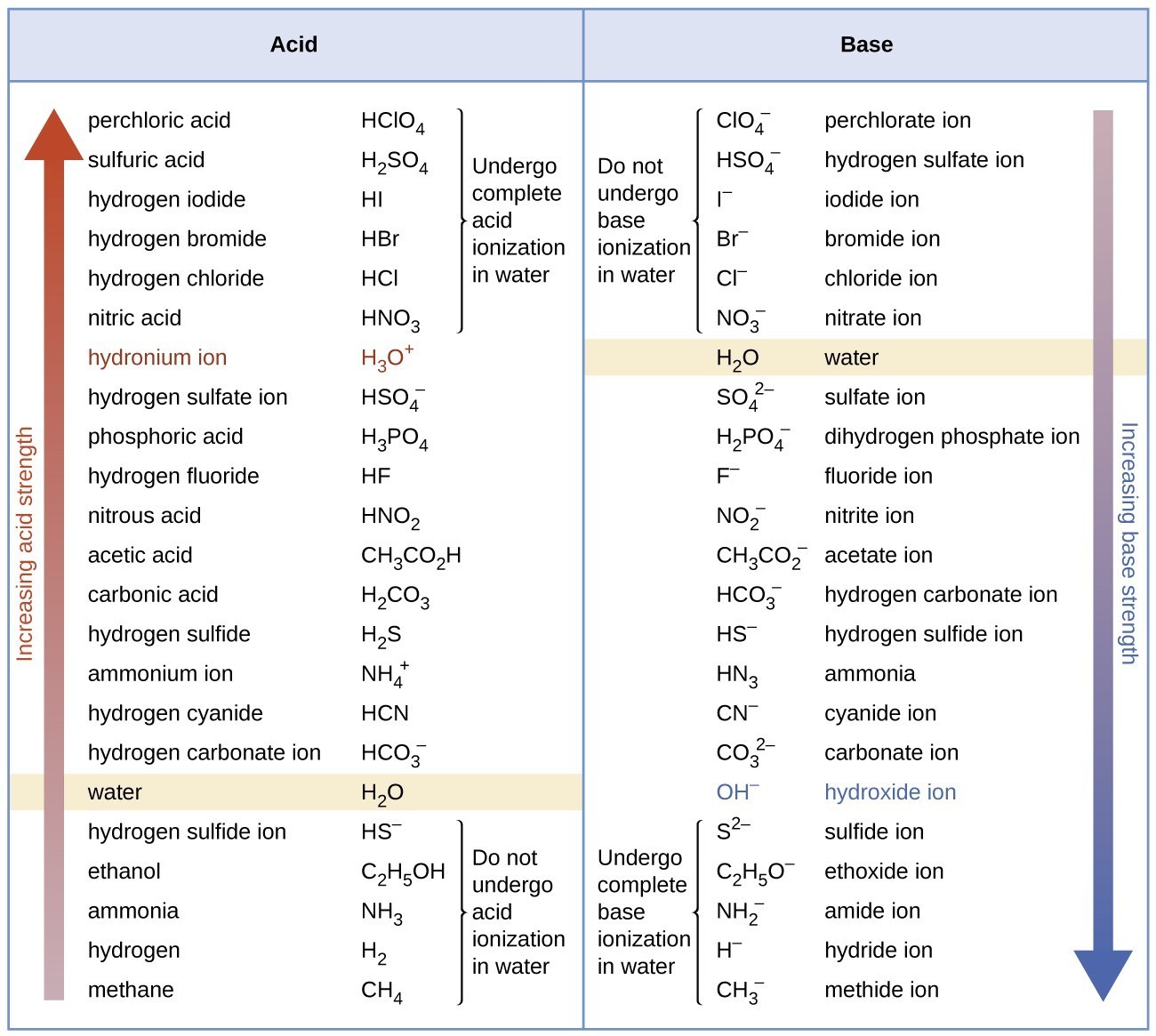
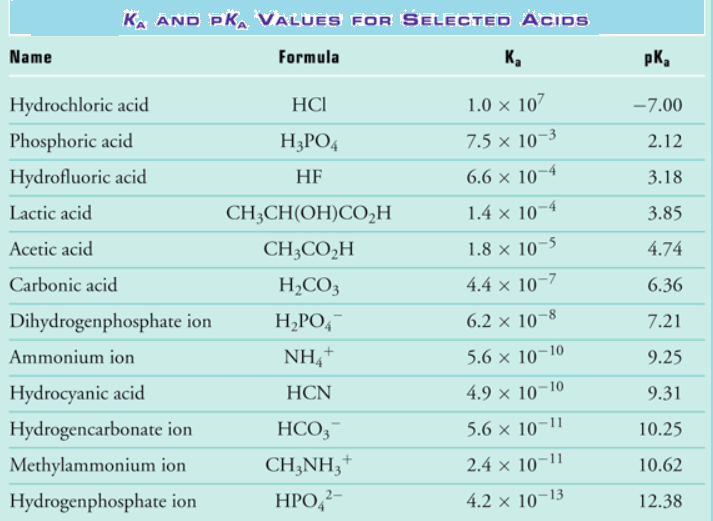
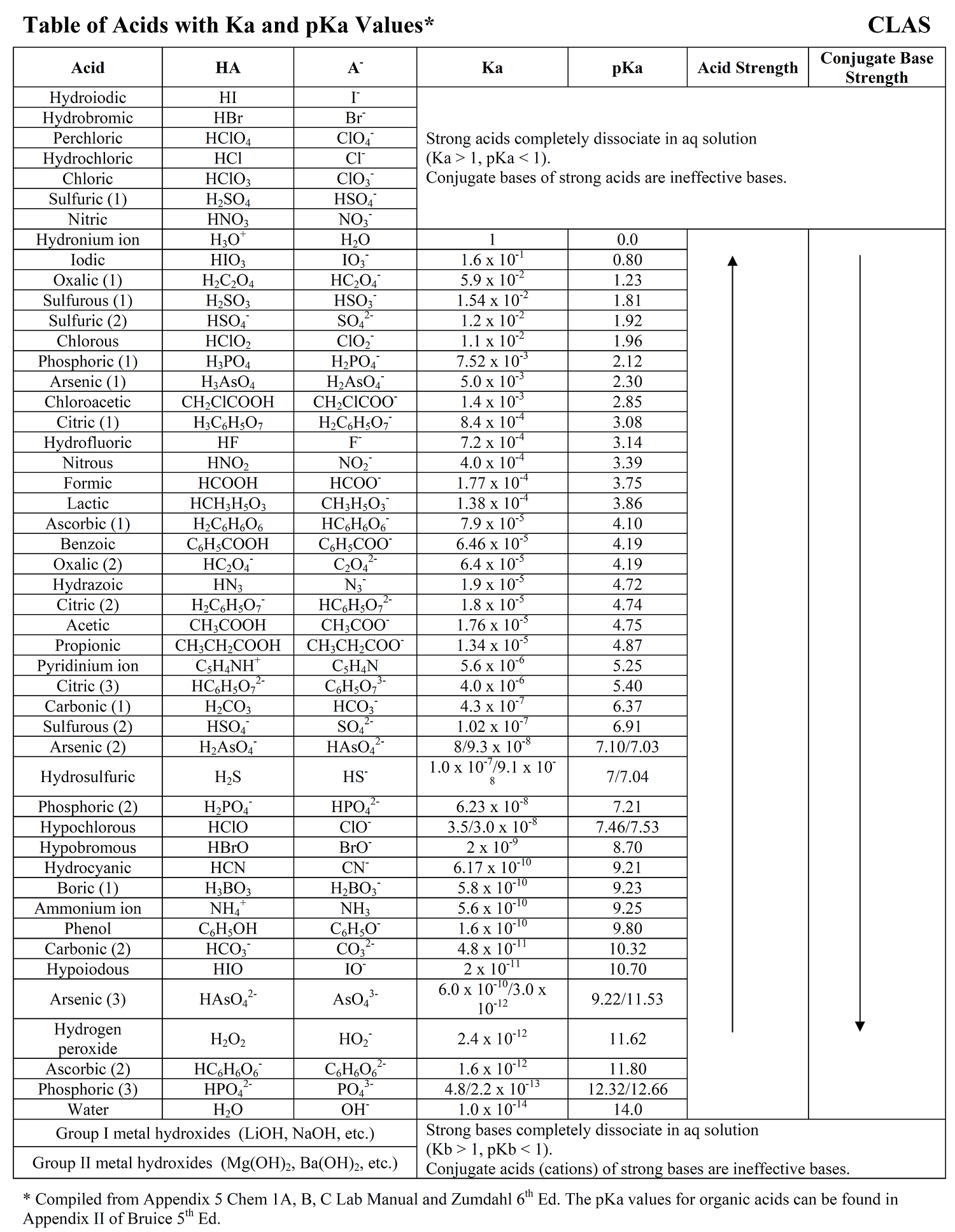
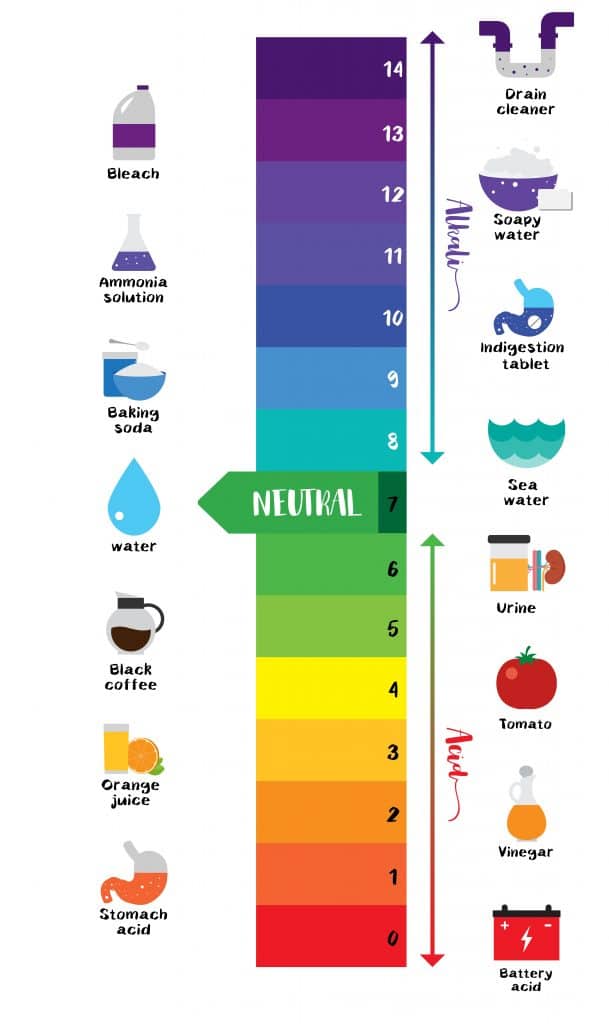
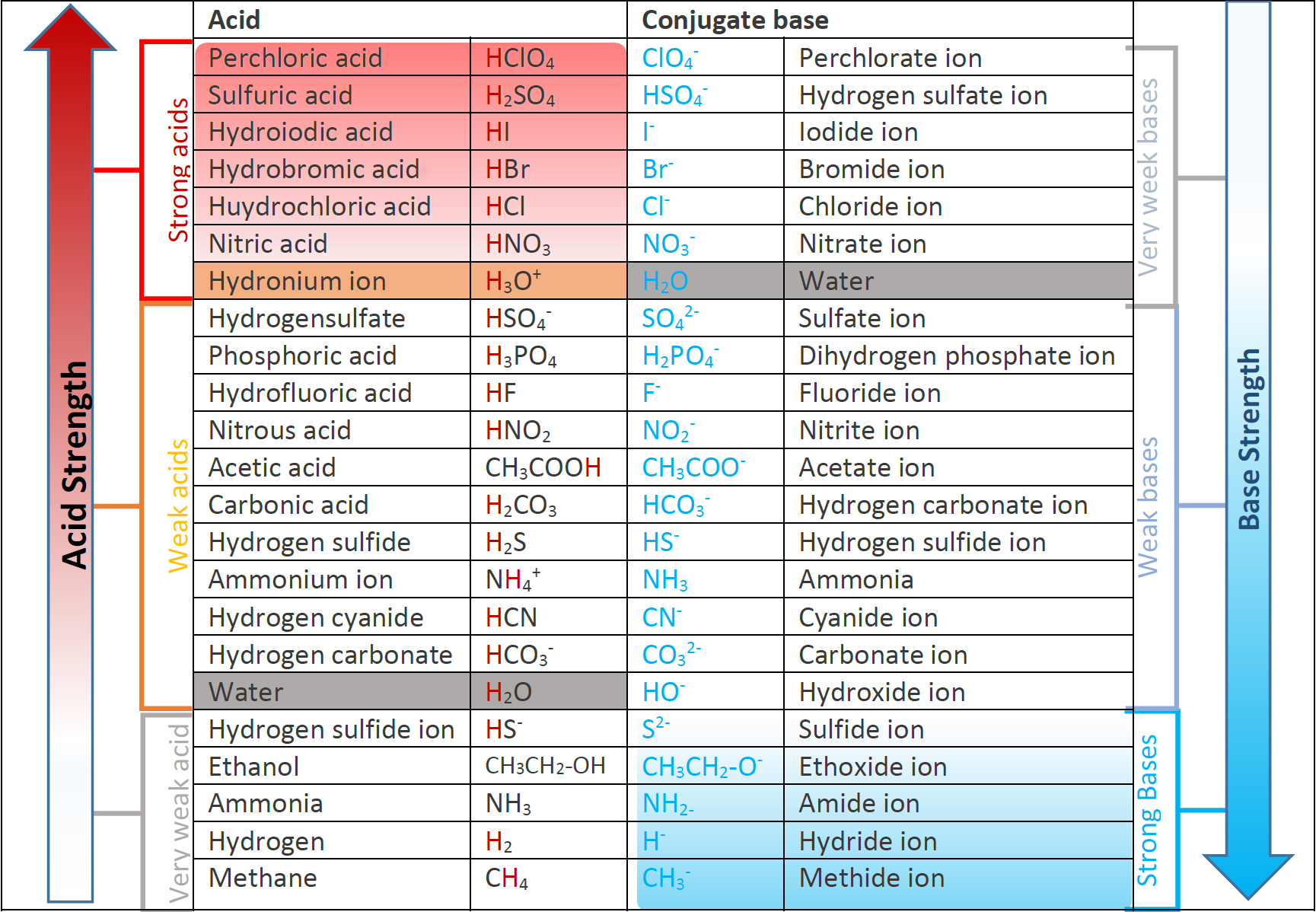
Acid Strength Chart
Acid Strength Chart - The relative strength of the acid/conjugate base pair is also. If relatively little ionization occurs, the acid or base is weak. Table \(\pageindex{1}\) lists several strong acids. Web the relative strength of an acid or base is the extent to which it ionizes when dissolved in water. The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions. Web use this acids and bases chart to find the relative strength of the most common acids and bases. Look at where the negative charge ends up in each conjugate base. Web figure 14.3.3 lists a series of acids and bases in order of the decreasing strengths of the acids and the corresponding increasing strengths of the bases. If the ionization reaction is essentially complete, the acid or base is termed strong; Web use this acids and bases chart to find the relative strength of the most common acids and bases. Web the terms strong and weak give an indication of the strength of an acid or base. Web chart of acid and base strength. If the ionization reaction is essentially complete, the acid or base is termed strong; A stronger acid (larger ka) has a smaller p ka, and a. Web use this acids and bases chart to find the relative strength of the most common acids and bases. For each acid, the ionization reaction shows the acid’s conjugate base. If relatively little ionization occurs, the acid or base is weak. Web a strong acid yields 100% (or very nearly so) of \(\ce{h3o+}\) and \(\ce{a^{−}}\) when the acid ionizes in water. Web use this acids and bases chart to find the relative strength of the most common acids and bases. A guide to acids, acid strength, and concentration. Examples of strong acids are hydrochloric acid. By andy brunning september 28, 2016. 48 × 32, each (ap7042). Web acid strength is the tendency of an acid, symbolised by the chemical formula, to dissociate into a proton, +, and an anion,. Web acid strengths are normally expressed using p ka values rather than ka values, where the pka is the. A guide to acids, acid strength, and concentration. The relative strength of the acid/conjugate base pair is also. The first six acids in figure 14.3.3 are the most common strong acids. Web acid with values less than one are considered weak. (select option to see volume pricing availability) 8½ × 11, pad of 30 (ap7229) $24.50. By andy brunning september 28, 2016. Web figure 14.3.3 lists a series of acids and bases in order of the decreasing strengths of the acids and the corresponding increasing strengths of the bases. The relationship between acid strength and the ph of a solution. The acid and base in a given row are conjugate to each other. Examples of strong. The relationship between acid strength and the ph of a solution. If the ionization reaction is essentially complete, the acid or base is termed strong; The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions. Examples of strong acids are hydrochloric acid. Web the terms strong and weak give an indication of the. Web the terms strong and weak give an indication of the strength of an acid or base. A stronger acid (larger ka) has a smaller p ka, and a. The strong bases are listed at the bottom right of the table and get weaker as we move to the top of the table. Web the relative strength of an acid. Web acid with values less than one are considered weak. If relatively little ionization occurs, the acid or base is weak. Web the strength of acids and bases, i.e., the extent of dissociation of the dissolved acid or base into ions in water is described. Chart or notebook size available. This information can be used to predict the outcome of. Examples of strong acids are hydrochloric acid. Pka = −log ka p k a = − log k a. For each acid, the ionization reaction shows the acid’s conjugate base. Web a strong acid yields 100% (or very nearly so) of \(\ce{h3o+}\) and \(\ce{a^{−}}\) when the acid ionizes in water. The first six acids in figure 15.3.3 are the most. Calculating the ph of a strong acid or base solution. Web acid with values less than one are considered weak. Web determine at a glance the relative strengths of a host of acids and bases. Web the strength of acids and bases, i.e., the extent of dissociation of the dissolved acid or base into ions in water is described. A. They’re routinely described as strong or weak, concentrated or dilute. Web use this acids and bases chart to find the relative strength of the most common acids and bases. Look at where the negative charge ends up in each conjugate base. Web acid strengths are normally expressed using p ka values rather than ka values, where the pka is the. Web use this acids and bases chart to find the relative strength of the most common acids and bases. Calculating the ph of a strong acid or base solution. The acid and base in a given row are conjugate to each other. The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions. Examples. Web the relative strength of an acid or base is the extent to which it ionizes when dissolved in water. Web the relative strength of an acid or base is the extent to which it ionizes when dissolved in water. A weak acid does not completely ionize in water, yielding only small amounts of \(\ce{h3o+}\) and \(\ce{a^{−}}\). The first six acids in figure 14.3.3 are the most common strong acids. By andy brunning september 28, 2016. The first six acids in figure 15.3.3 are the most common strong acids. A stronger acid (larger ka) has a smaller p ka, and a. Web a reactivity series, or acid strength chart, provides a visual representation of the relative strength of various acids. Examples of strong acids are hydrochloric acid. The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions. Web figure 14.3.3 lists a series of acids and bases in order of the decreasing strengths of the acids and the corresponding increasing strengths of the bases. If the acid or base conducts electricity strongly, it is a strong acid or base. For each acid, the ionization reaction shows the acid’s conjugate base. Acid strength increases down a group and increases from left to right across a period. A guide to acids, acid strength, and concentration. Chart or notebook size available.List of Strong Acids & Bases in Order StudyPK
Acid Strengths Table
Relative Strengths of Acids and Bases Chemistry Atoms First
Acid strength W3schools
Acid Strength, Ka, and pKa Chemistry Steps
Section 3 Strengths of Acids and Bases Nitty Gritty Science
6.3 Strength of acids and bases Chemistry LibreTexts
pKa Values and strengths of Acids and Bases
AcidBase Strength Charts for Chemistry
14.3 Relative Strengths of Acids and Bases Chemistry LibreTexts
Web Definitions Of Ph, Poh, And The Ph Scale.
Web Acid Strengths Are Normally Expressed Using P Ka Values Rather Than Ka Values, Where The Pka Is The Negative Common Logarithm Of The Ka:
Calculating The Ph Of A Strong Acid Or Base Solution.
The More Stable (Weaker) The Conjugate Base, The Stronger The Acid.
Related Post: