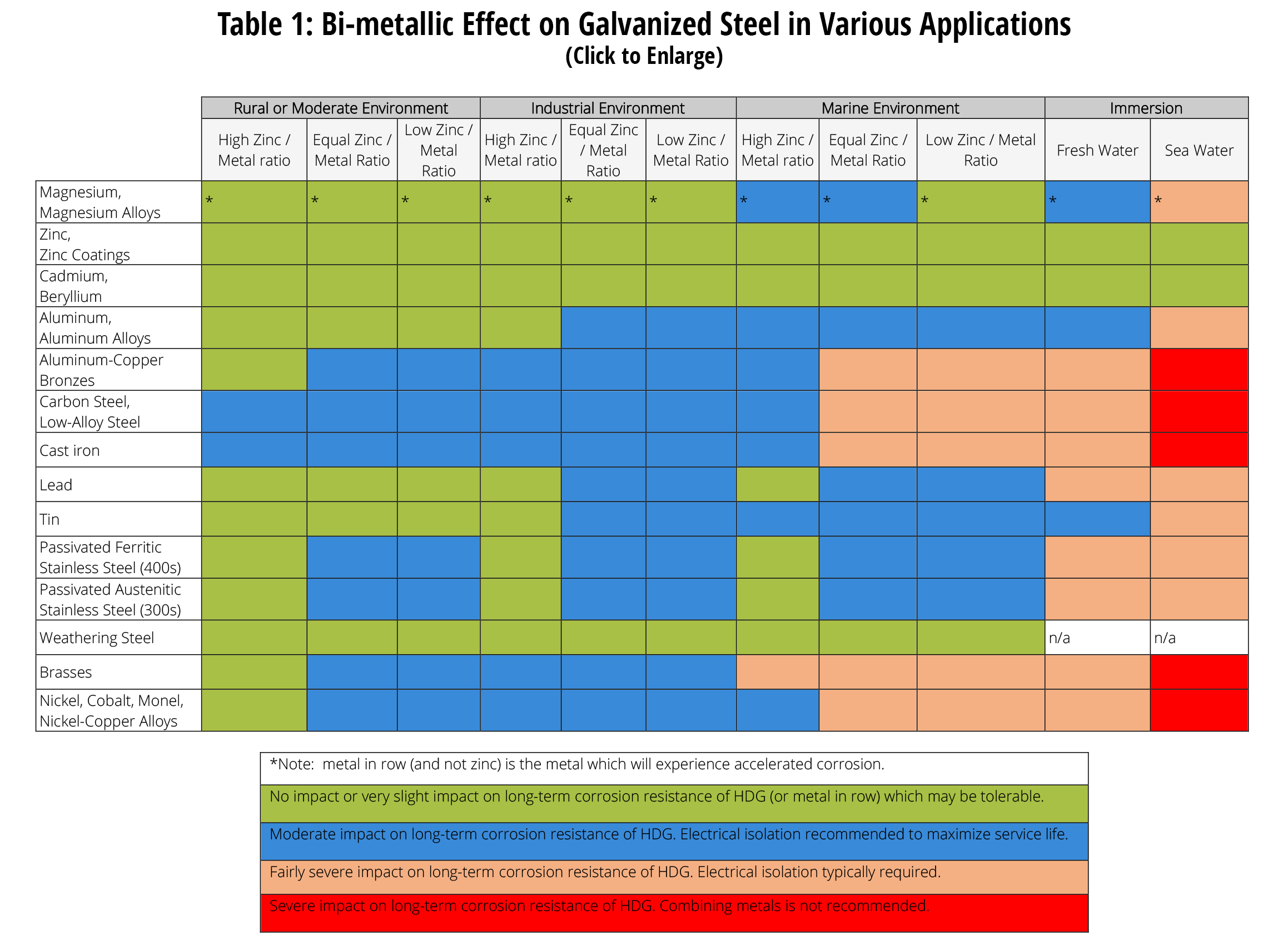
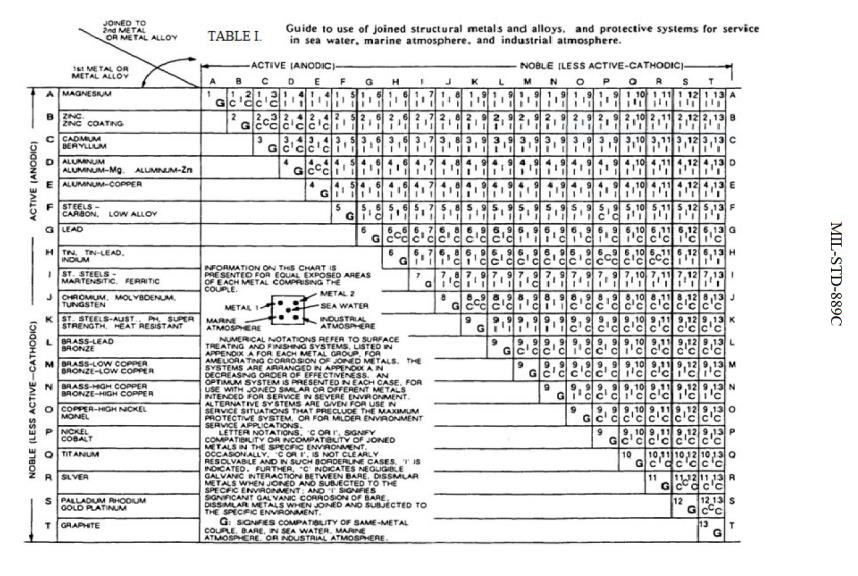
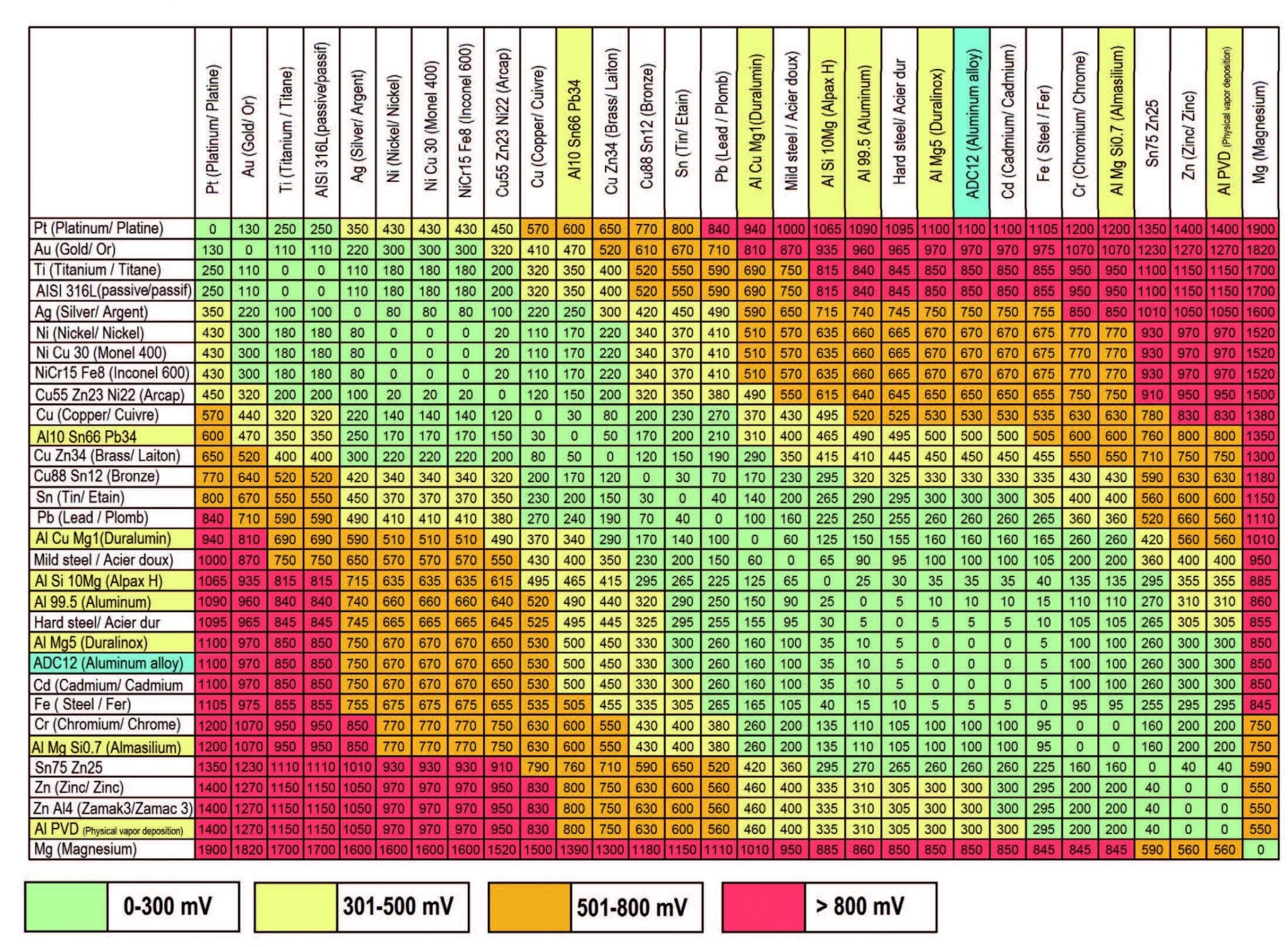
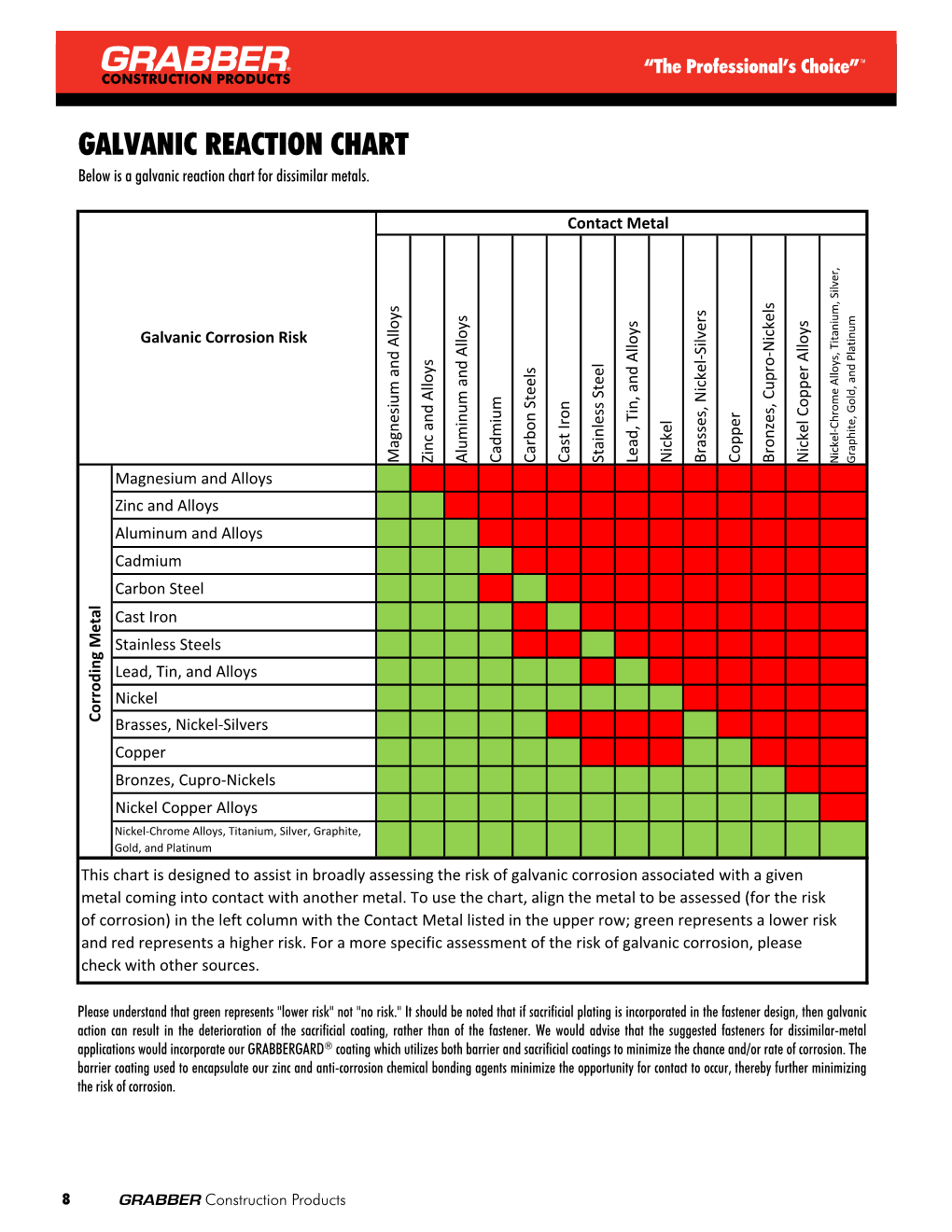
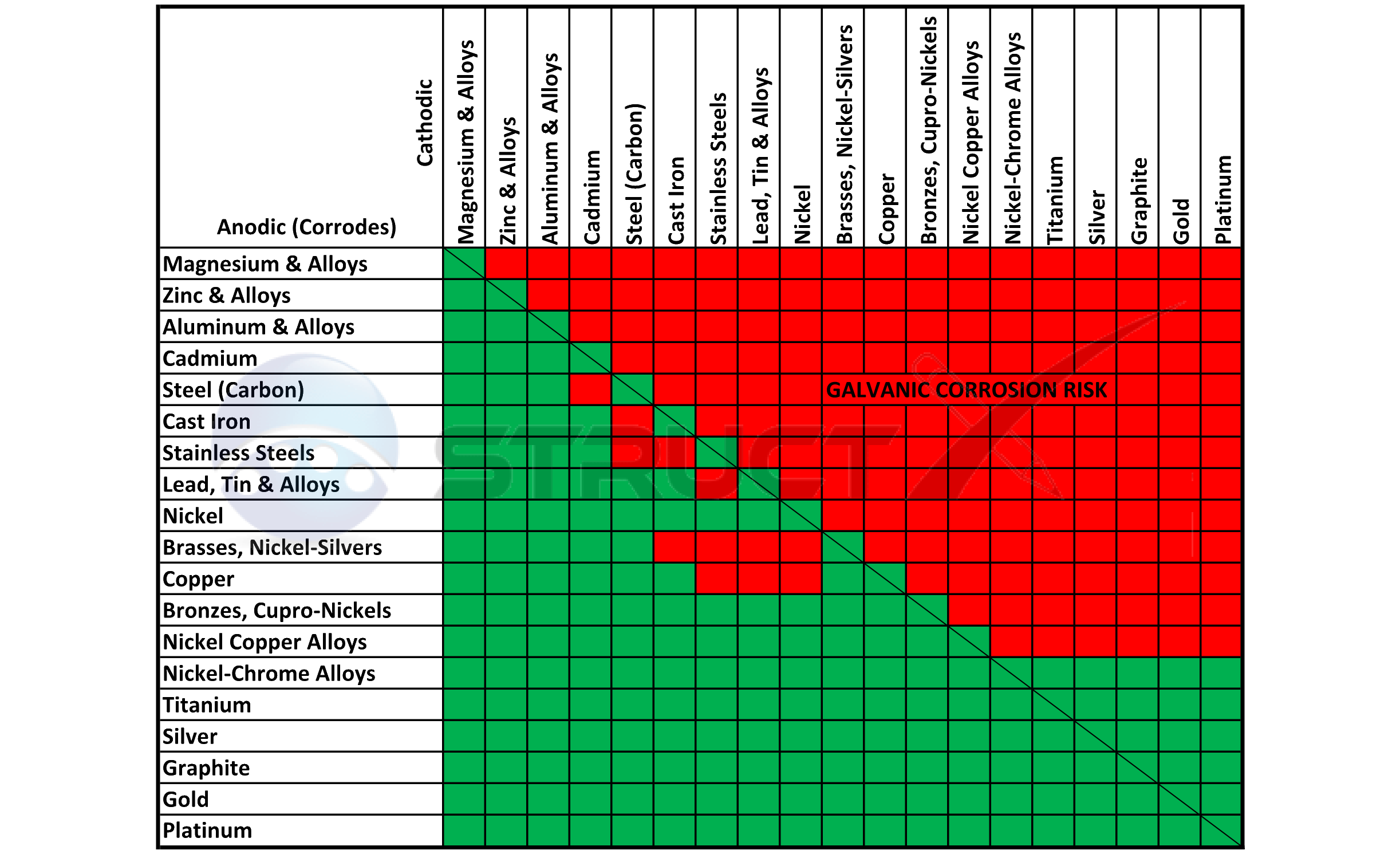
Dissimilar Metals Chart
Dissimilar Metals Chart - This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Web galvanic or dissimilar metal corrosion is electrochemical corrosion that occurs when one metal comes in contact with another material. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. When dissimilar metals are used together in the presence of an electrolyte, separate them with a dielectric material such as insulation, paint or similar surface coating. A typical rule of thumb is that voltage differences of 0.2 volts or more suggest a galvanic corrosion risk. Web our extensive testing and independent certification program provide our customers access to over 40 florida product approvals on 11 different metal panel profiles. The cart to the left is galvanic series in flowing sea water. This conversion resource can help you determine what metal gauge you may need. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). When dissimilar metals are used together in the presence of an electrolyte, separate them with a dielectric material such as insulation, paint or similar surface coating. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal combinations. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). Weathertight warranties are also available. When working with copper or aluminum use antioxidant pastes. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. Web below is a galvanic reaction chart for dissimilar metals. Web this article examines how dissimilar metals can lead to galvanic corrosion. Web below is a galvanic reaction chart for dissimilar metals. Web galvanic corrosion potential is a measure of how dissimilar metals will corrode when placed against each other in an assembly. We also offer technical support including easy access to installation guides, product literature, technical bulletins and color charts. Web galvanic corrosion occurs when two dissimilar metals with different potentials are placed in electrical contact in an electrolyte. Web choosing the right size or area of the joined metals: Web when two different metals are in contact and exposed to a. Web chart sheet metal gauge. There are three conditions that must exist for galvanic corrosion to occur. It includes a chart that shows how different plating materials react to one another with regard to their galvanic potential. Weathertight warranties are also available. Web galvanic or dissimilar metal corrosion is electrochemical corrosion that occurs when one metal comes in contact with. Web this article examines how dissimilar metals can lead to galvanic corrosion. This type of accelerated corrosion between dissimilar metals is referred to as galvanic corrosion. Web we are aa metals, inc, one of north america’s largest master distributors of quality aluminum and stainless products. Web galvanic corrosion (some times called dissimilar metal corrosion) is the process by which the. We also provide other helpful methods for avoiding galvanic corrosion. When dissimilar metals are used together in the presence of an electrolyte, separate them with a dielectric material such as insulation, paint or similar surface coating. Web but when you step onto a jobsite, working with dissimilar metals turns into a whole new monster with dire potential consequences. • harsh. Web galvanic corrosion potential is a measure of how dissimilar metals will corrode when placed against each other in an assembly. First there must be two electrochemically dissimilar metals present. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). The cart to the left is galvanic series in flowing sea. • harsh environments, such as outdoors, high humidity, and salt environments. The greater the potential difference is, the greater the tendency for corrosion. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. First there must be two electrochemically dissimilar metals present. Web this slide. Web when two different metals are in contact and exposed to a common electrolyte, one of the metals experiences accelerated corrosion while the other is protected. The cart to the left is galvanic series in flowing sea water. Web this slide includes a chart of galvanic corrosion potential between common construction metals. When dissimilar metals are used together in the. This type of accelerated corrosion between dissimilar metals is referred to as galvanic corrosion. Web read on to find out about what it is and how to use it to analyse the compatibility of joining metals. When working with copper or aluminum use antioxidant pastes. Web but when you step onto a jobsite, working with dissimilar metals turns into a. In this article, we'll look at an example to illustrate the use of the galvanic table. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. Web this slide includes a chart of galvanic corrosion potential between common construction metals. A typical rule of thumb. Web below is a galvanic reaction chart for dissimilar metals. • harsh environments, such as outdoors, high humidity, and salt environments. Electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amounts of acid. Web below is a galvanic reaction chart for dissimilar metals. Web choosing the. Electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing very small amounts of acid. Web galvanic corrosion potential is a measure of how dissimilar metals will corrode when placed against each other in an assembly. Web this slide includes a chart of galvanic corrosion potential between common construction metals. Web galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. In this article, we'll look at an example to illustrate the use of the galvanic table. This conversion resource can help you determine what metal gauge you may need. • harsh environments, such as outdoors, high humidity, and salt environments. Web but when you step onto a jobsite, working with dissimilar metals turns into a whole new monster with dire potential consequences. Web the galvanic series chart below shows metals and their electrochemical voltage range (relative activity in flowing sea water). The cart to the left is galvanic series in flowing sea water. A typical rule of thumb is that voltage differences of 0.2 volts or more suggest a galvanic corrosion risk. There are three conditions that must exist for galvanic corrosion to occur. Web we are aa metals, inc, one of north america’s largest master distributors of quality aluminum and stainless products. Weathertight warranties are also available. First there must be two electrochemically dissimilar metals present. When working with copper or aluminum use antioxidant pastes.Dissimilar Metal Corrosion with… American Galvanizers Association
Dissimilar Welding Chart Stainless Steel Transition Metals
Dissimilar metal corrosion with chemical filmed (Alodine, Iridite
Galvanic Corrosion Chart Dissimilar Metals A Visual Reference of
Galvanic Reaction Chart
Dissimilar Metals Corrosion Chart
Galvanic Corrosion Chart Dissimilar Metals
Dissimilar Metal Corrosion Chart
Dissimilar joining of Al with steel? r/Welding
Design Calculations of Lightning Protection Systems Part Fifteen
Web When Two Different Metals Are In Contact And Exposed To A Common Electrolyte, One Of The Metals Experiences Accelerated Corrosion While The Other Is Protected.
This Chart Is Designed To Assist In Broadly Assessing The Risk Of Galvanic Corrosion Associated With A Given Metal Coming Into Contact With Another Metal.
Web Galvanic Corrosion Occurs When Two Dissimilar Metals With Different Potentials Are Placed In Electrical Contact In An Electrolyte.
You Can Also Learn More About Overcoming Potentially Compatibility Issues Between Metals.
Related Post: