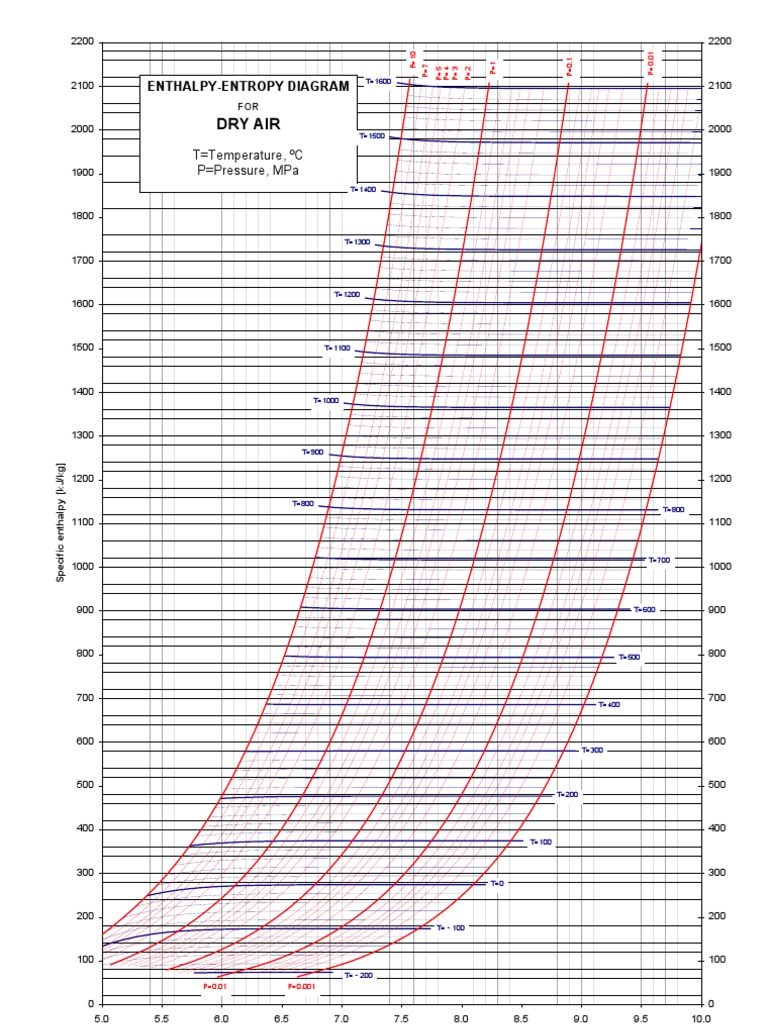
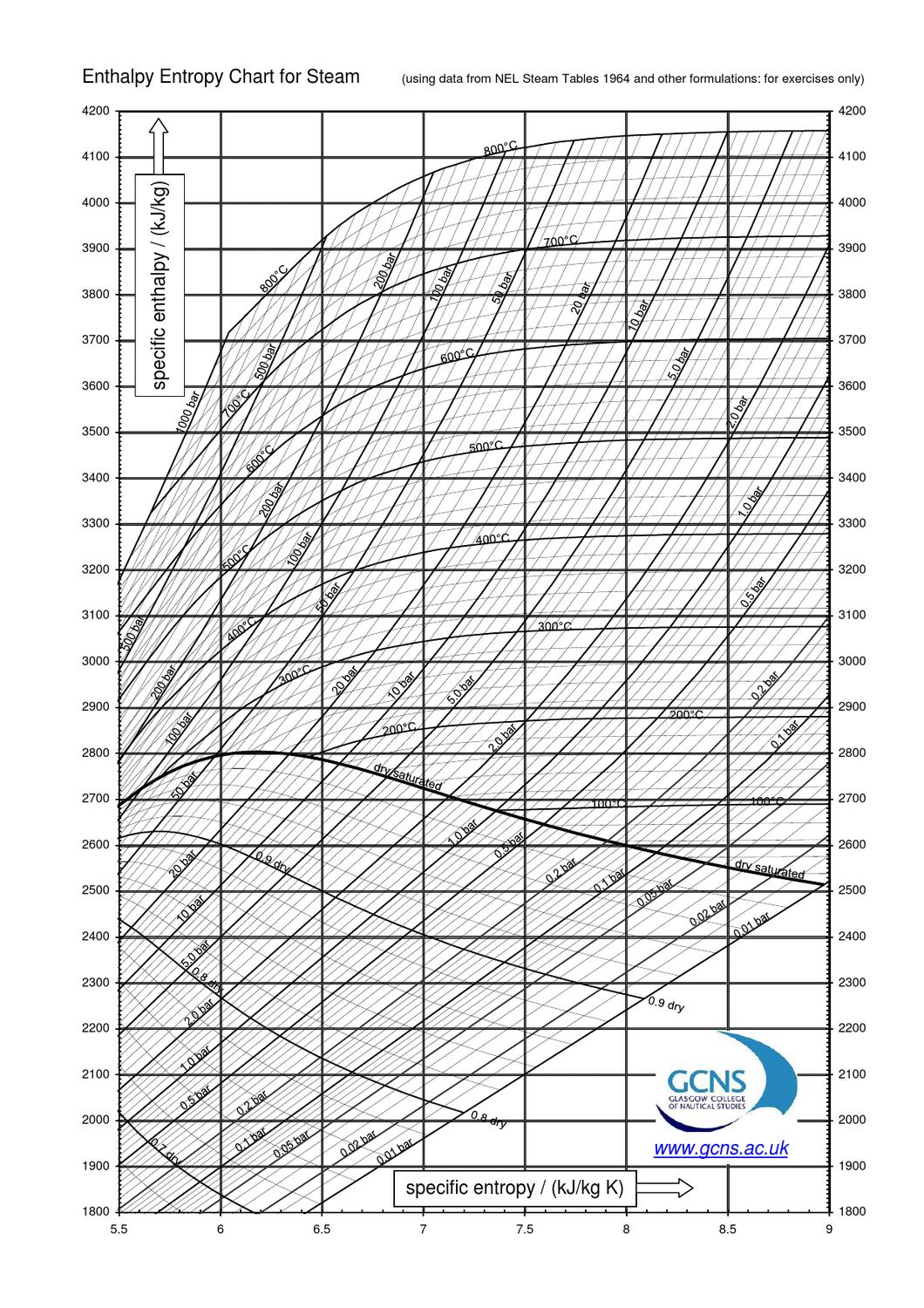
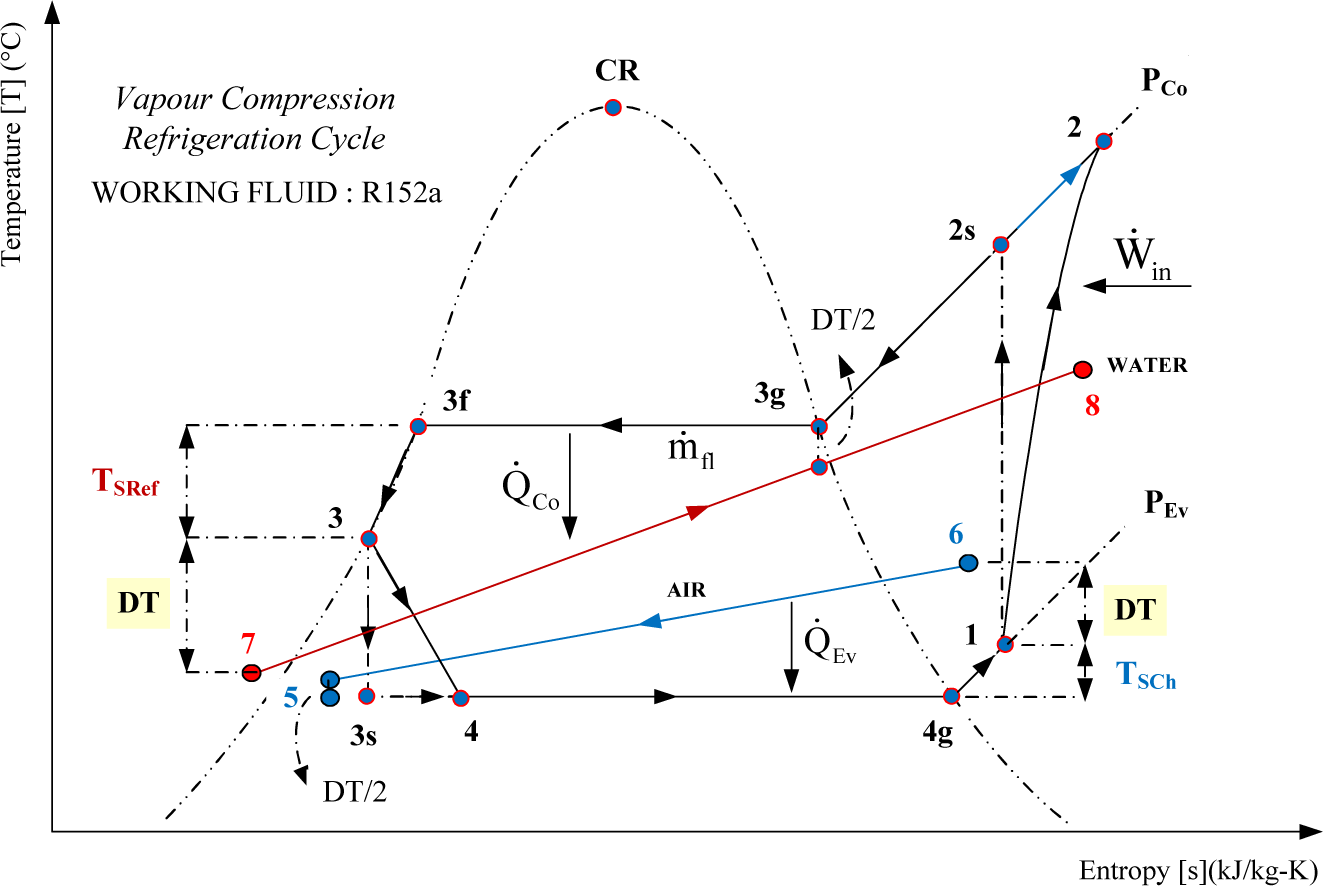
Enthalpy Entropy Chart
Enthalpy Entropy Chart - New york, new york, 1979; When energy is added) or decreases. Enthalpy is defined as the total heat content or total useful energy of a substance. Web the heat that passes into or out of the system during a reaction is the enthalpy change. U = specific internal energy. Web standardized enthalpies and entropies of some common substances: Web the mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy of. The messy room on the right has more. The mollier diagram is useful. Go to standard state and standard enthalpy of formation for. S=0 kj/k*kg and h=0 kj/kg. Web steam tables online calculator, completely free! Web the figures and tables below shows how water enthalpy and entropy changes with temperature (°c and °f) at water saturation pressure (which for practicle use, gives the. U = specific internal energy. Web based on the phase diagram (supplementary fig. Web standardized enthalpies and entropies of some common substances: When energy is added) or decreases. Whether the enthalpy of the system increases (i.e. Some important terms related to enthalpy: New york, new york, 1979; Calculate propierties of wet, saturated and superheated steam, steam quality and more. Web the mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy of. As was discussed earlier in this text, natural processes tend towards increasing entropy. Web the mollier diagram is a chart on which enthalpy (h) versus. Web the mollier diagram is a chart on which enthalpy (h) versus entropy (s) is plotted. New york, new york, 1979; Calculate propierties of wet, saturated and superheated steam, steam quality and more. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. S=0 kj/k*kg and h=0 kj/kg. Some important terms related to enthalpy: The symbol for enthalpy is “h.” enthalpy is also considered to be the sum of. Enthalpy is defined as the total heat content or total useful energy of a substance. When energy is added) or decreases. S=0 kj/k*kg and h=0 kj/kg. Understanding enthalpy and entropy clarify what these. Calculate propierties of wet, saturated and superheated steam, steam quality and more. Web definition and explanation of the terms standard state and standard enthalpy of formation, with listing of values for standard enthalpy and gibbs free energy of formation, as well. New york, new york, 1979; Web enthalpy ( / ˈɛnθəlpi / ⓘ). The messy room on the right has more. As was discussed earlier in this text, natural processes tend towards increasing entropy. Go to standard state and standard enthalpy of formation for. It is a state function in thermodynamics used in. Equilibrium states at 25oc (77of) and 1 atm. New york, new york, 1979; Calculate propierties of wet, saturated and superheated steam, steam quality and more. S=0 kj/k*kg and h=0 kj/kg. Some important terms related to enthalpy: As was discussed earlier in this text, natural processes tend towards increasing entropy. When energy is added) or decreases. Enthalpy is defined as the total heat content or total useful energy of a substance. As was discussed earlier in this text, natural processes tend towards increasing entropy. It is a state function in thermodynamics used in. Whether the enthalpy of the system increases (i.e. Web enthalpy ( / ˈɛnθəlpi / ⓘ) is the sum of a thermodynamic system 's internal energy and the product of its pressure and volume. The mollier diagram is useful. Equilibrium states at 25oc (77of) and 1 atm. When energy is added) or decreases. Web the diagram below can be used to determine enthalpy versus entropy of water and steam. Go to standard state and standard enthalpy of formation for. U = specific internal energy. Some important terms related to enthalpy: What is the enthalpy change? Enthalpy is defined as the total heat content or total useful energy of a substance. S=0 kj/k*kg and h=0 kj/kg. Whether the enthalpy of the system increases (i.e. Calculate propierties of wet, saturated and superheated steam, steam quality and more. As was discussed earlier in this text, natural processes tend towards increasing entropy. Web the mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy. It is a state function in thermodynamics used in. Web enthalpy ( / ˈɛnθəlpi / ⓘ) is the sum of a thermodynamic system 's internal energy and the product of its pressure and volume. The mollier diagram is useful. Web the mollier diagram is a chart on which enthalpy (h) versus entropy (s) is plotted. Equilibrium states at 25oc (77of) and 1 atm. Go to standard state and standard enthalpy of formation for. The messy room on the right has more. Web the heat that passes into or out of the system during a reaction is the enthalpy change. S=0 kj/k*kg and h=0 kj/kg. Web the figures and tables below shows how water enthalpy and entropy changes with temperature (°c and °f) at water saturation pressure (which for practicle use, gives the. U = specific internal energy. The symbol for enthalpy is “h.” enthalpy is also considered to be the sum of. What is the enthalpy change? Calculate propierties of wet, saturated and superheated steam, steam quality and more. Web the mollier diagram is a graph used in thermodynamics to visualize the relationships between temperature, pressure, specific volume, enthalpy, and entropy of. Whether the enthalpy of the system increases (i.e.Entropy And Enthalpy Chart
Gibbs Free Energy Entropy, Enthalpy & Equilibrium Constant K YouTube
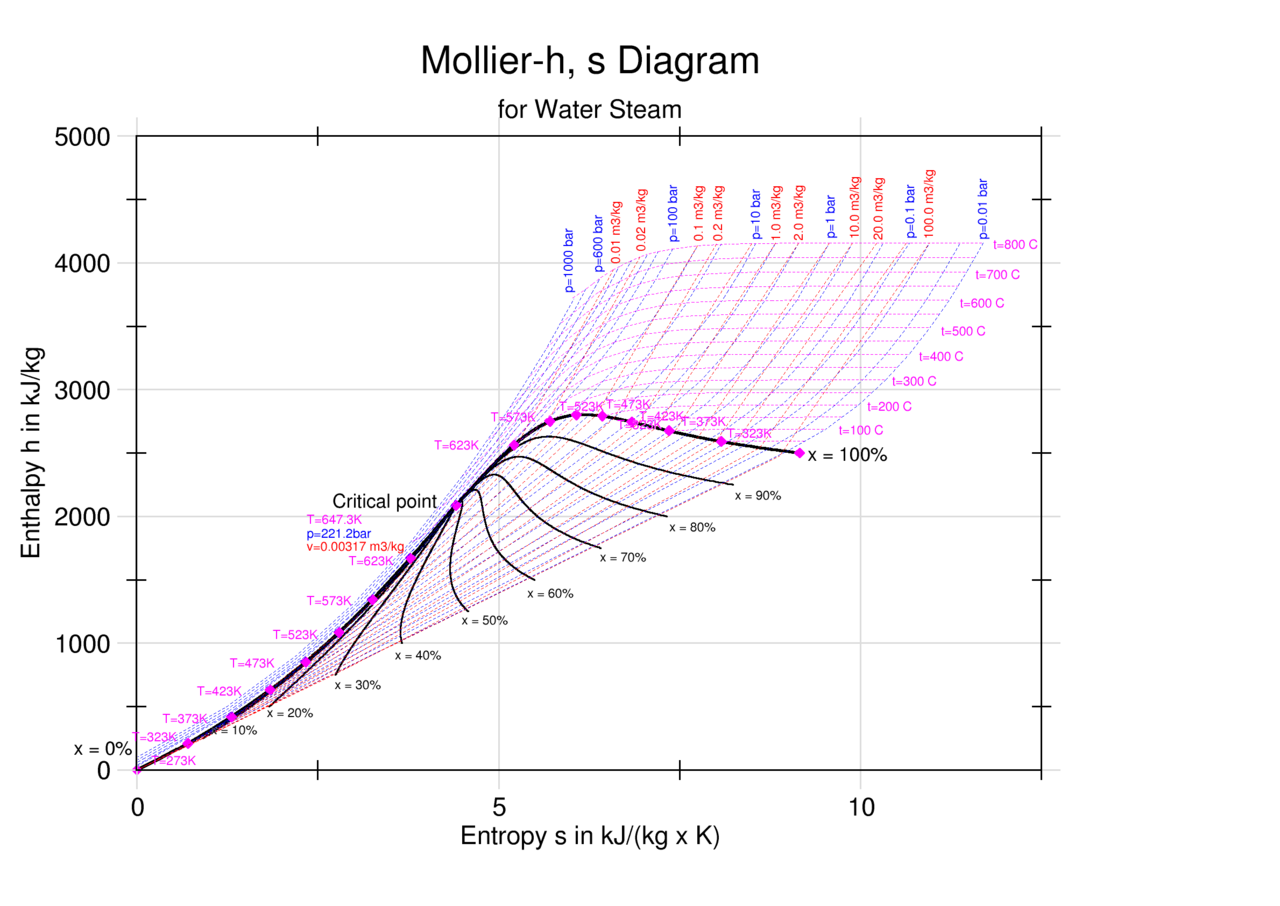
Enthalpy And Entropy Chart
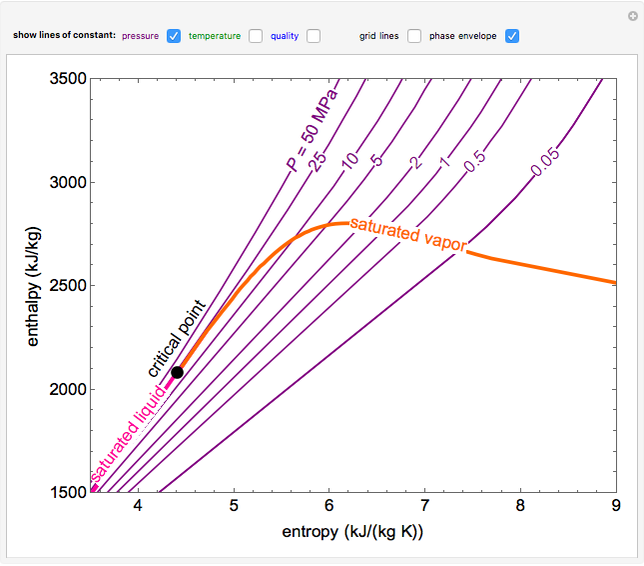
EnthalpyEntropy Diagram for Water Wolfram Demonstrations Project
Enthalpy Chart Chemistry
Entropy And Enthalpy Chart
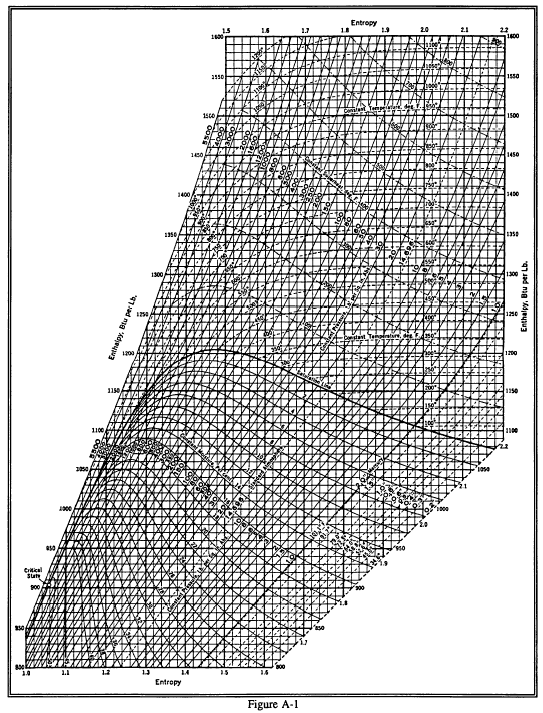
Enthalpy Entropy (hs) or Mollier Diagram Engineers Edge www
EnthalpyEntropy Diagram for Water Wolfram Demonstrations Project
Enthalpy Entropy (hs) or Mollier Diagram
Enthalpy And Entropy Chart
Web Where Enthalpy Is A Measurement Of Energy Potential, Entropy Measures The Randomness Of Energy With Relation To Heat.
Some Important Terms Related To Enthalpy:
New York, New York, 1979;
Web Standardized Enthalpies And Entropies Of Some Common Substances:
Related Post: