Polarity Chart For Solvents
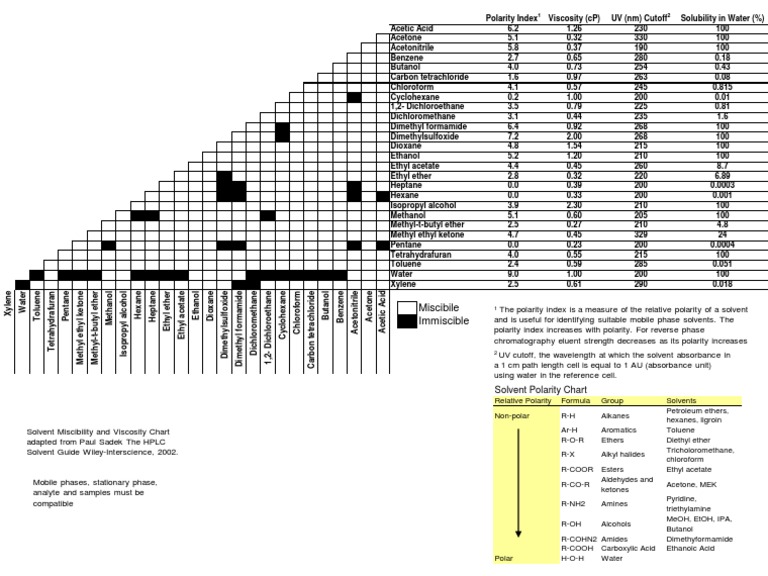
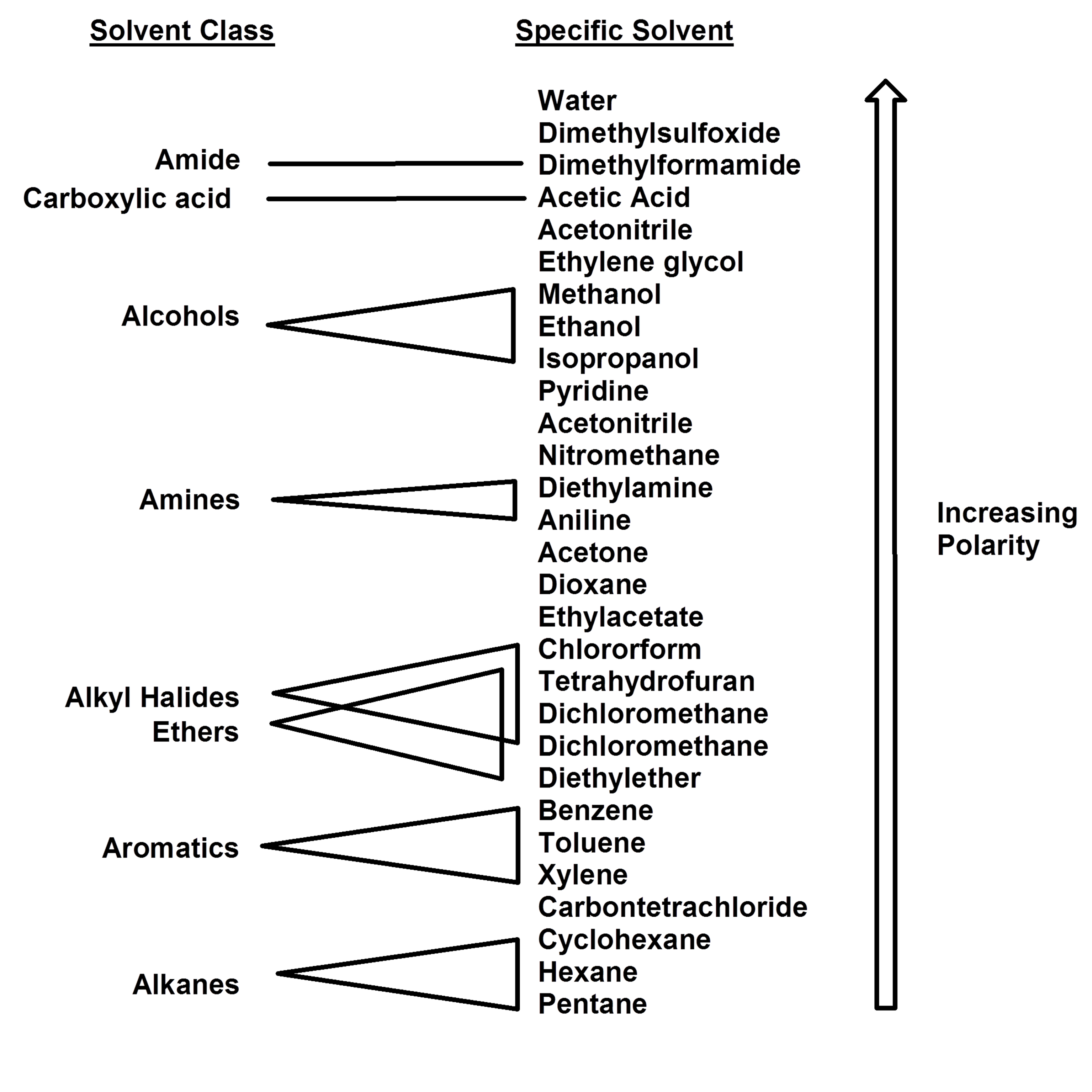
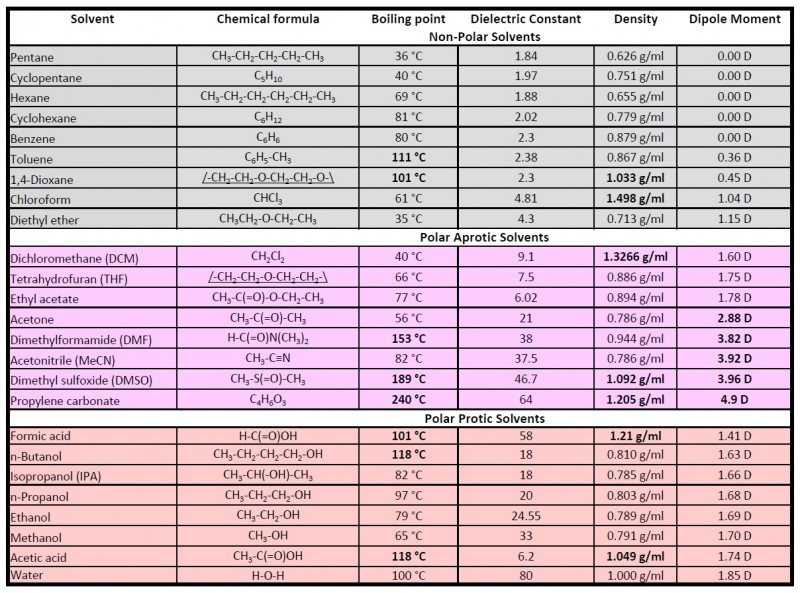
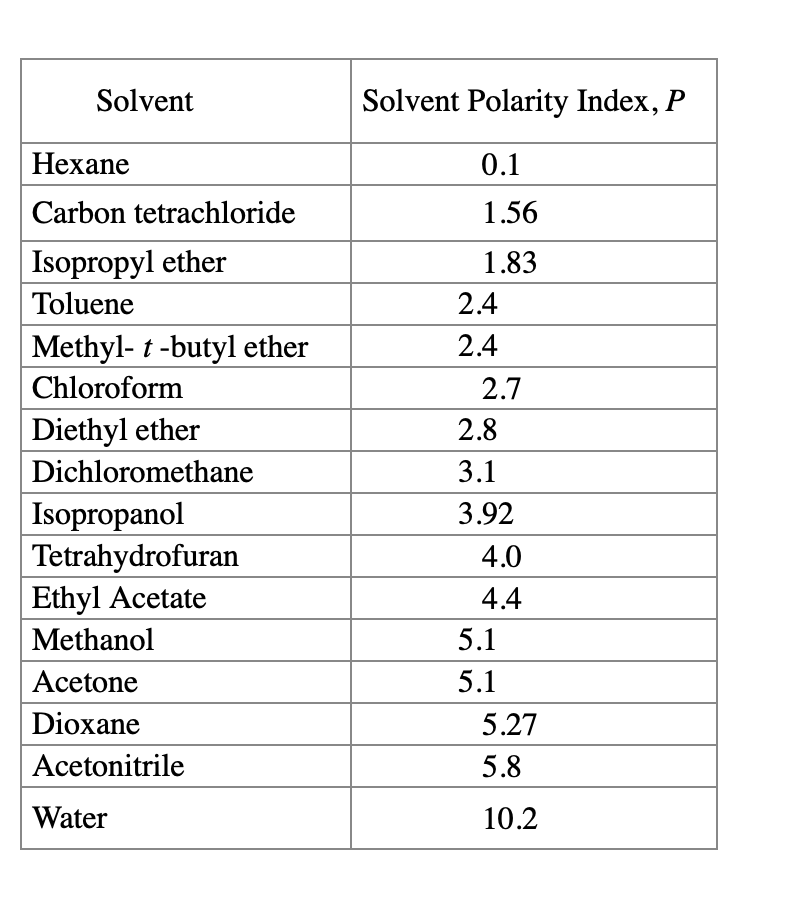
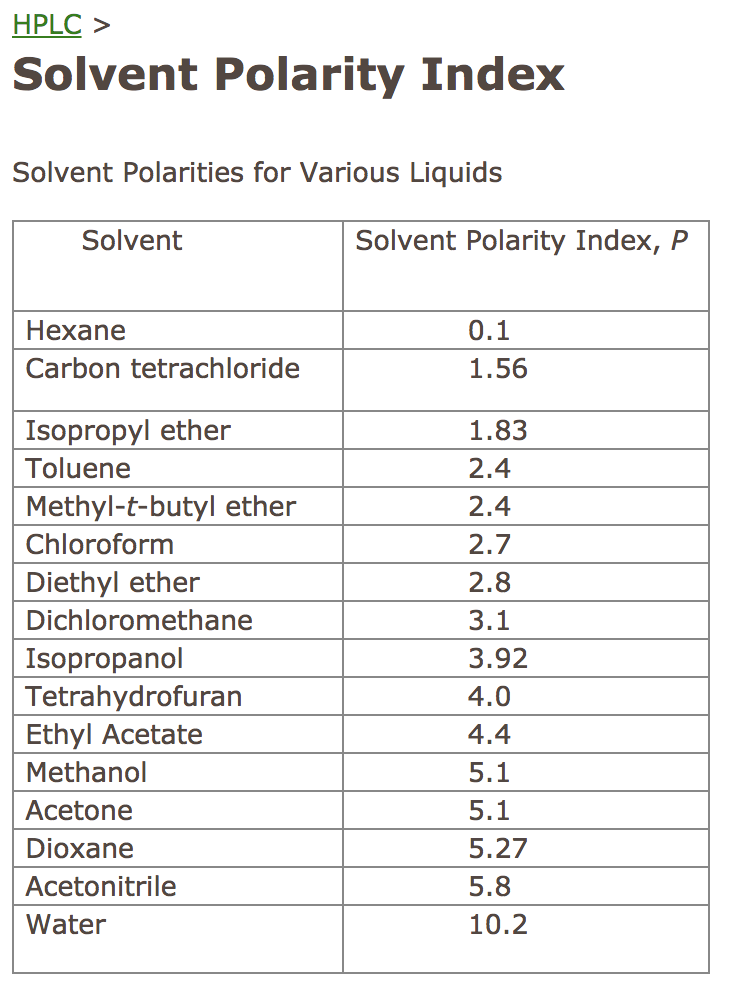
Polarity Chart For Solvents - Assess the polarity of a molecule based on its bonding and structure Web this tool allows you interactively select solvents based upon the principal component analysis (pca) of the solvent's physical properties. 641 shows some of the most common crystallization solvents arranged by order of decreasing polarity going from top to bottom. Web explain the concepts of polar covalent bonds and molecular polarity; Web because polar solvents stabilize the ground state more than the excited state, the change in energy increases with the polarity of the solvent. Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying suitable mobile phase solvents. Web 47 rows information on the properties of common solvents used in organic chemistry including boiling points, solubility, density, dielectric constants, and flash points. Web the document lists various solvents and provides their polarity index, viscosity, uv cutoff wavelength, solubility in water, and miscibility. Demystifying synthetic organic chemistry since 2004. Laboratory techniques and methods to. This page uses frames, but your browser doesn't support them. Web explain the concepts of polar covalent bonds and molecular polarity; It also includes charts on relative solvent. Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying suitable mobile phase solvents. Web the polarity of a covalent bond can be judged by determining the difference of the electronegativities of the two atoms involved in the covalent bond, as summarized. Web 23 rows solvent polarity; 641 shows some of the most common crystallization solvents arranged by order of decreasing polarity going from top to bottom. Demystifying synthetic organic chemistry since 2004. Common solvents arranged from the least polar to the most polar Web 24 rows polarity indexes of solvents which are commonly used for sec analysis is. The values for relative polarity are normalized from measurements of solvent shifts of absorption spectra and were extracted from christian reichardt,. Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying suitable mobile phase solvents. Assess the polarity of a molecule based on its bonding and structure Water acetic acid. Web properties of solvents used in organic chemistry including mp, bp, desnity, water solubiity, polarity viscosity, dipole moment, dielectric constant Web the document lists various solvents and provides their polarity index, viscosity, uv cutoff wavelength, solubility in water, and miscibility. Demystifying synthetic organic chemistry since 2004. Laboratory techniques and methods to. Web burdick & jackson solvents are arranged in order. Solvents which are close to each. These ‘‘normalized selectivity’’ properties recognize three contributions of the solvent to. However, novel and new substances or solvent combinations are. This column is based on one of the most widely used polar phases, carbowax™ 20m, and is a polar column suitable for analyses of solvents, fatty acid. Water acetic acid ethylene glycol methanol ethanol. Web because polar solvents stabilize the ground state more than the excited state, the change in energy increases with the polarity of the solvent. P1 increases with solvent polarity. Water acetic acid ethylene glycol methanol ethanol isopropanol pyridine acetonitrile nitromethane diethylamine aniline dimethylsulfoxide ethyl acetate. This column is based on one of the most widely used polar phases, carbowax™ 20m,. Web because polar solvents stabilize the ground state more than the excited state, the change in energy increases with the polarity of the solvent. This column is based on one of the most widely used polar phases, carbowax™ 20m, and is a polar column suitable for analyses of solvents, fatty acid. Common solvents arranged from the least polar to the. Web 47 rows information on the properties of common solvents used in organic chemistry including boiling points, solubility, density, dielectric constants, and flash points. Web today, we will explore how polar organic solvents are, and will test what mixes with what. Web it's easy enough to look up the miscibility of common laboratory solvents on charts like the one provided. Web the document lists various solvents and provides their polarity index, viscosity, uv cutoff wavelength, solubility in water, and miscibility. Web 24 rows polarity indexes of solvents which are commonly used for sec analysis is. Demystifying synthetic organic chemistry since 2004. Web 23 rows solvent polarity; Web the polarity of a covalent bond can be judged by determining the difference. Web it's easy enough to look up the miscibility of common laboratory solvents on charts like the one provided below; Assess the polarity of a molecule based on its bonding and structure 641 shows some of the most common crystallization solvents arranged by order of decreasing polarity going from top to bottom. Web explain the concepts of polar covalent bonds. Web explain the concepts of polar covalent bonds and molecular polarity; Web this tool allows you interactively select solvents based upon the principal component analysis (pca) of the solvent's physical properties. This page uses frames, but your browser doesn't support them. Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for. Web this tool allows you interactively select solvents based upon the principal component analysis (pca) of the solvent's physical properties. Water acetic acid ethylene glycol methanol ethanol isopropanol pyridine acetonitrile nitromethane diethylamine aniline dimethylsulfoxide ethyl acetate. Web it's easy enough to look up the miscibility of common laboratory solvents on charts like the one provided below; Web 47 rows information. Some compounds are clearly very polar (e.g. P1 increases with solvent polarity. These ‘‘normalized selectivity’’ properties recognize three contributions of the solvent to. This page uses frames, but your browser doesn't support them. Web 1 the polarity index is a measure of the relative polarity of a solvent and is useful for identifying suitable mobile phase solvents. Assess the polarity of a molecule based on its bonding and structure Web table 10.1 on p. The values for relative polarity are normalized from measurements of solvent shifts of absorption spectra and were extracted from christian reichardt,. Water acetic acid ethylene glycol methanol ethanol isopropanol pyridine acetonitrile nitromethane diethylamine aniline dimethylsulfoxide ethyl acetate. Web 24 rows polarity indexes of solvents which are commonly used for sec analysis is. Web the document lists various solvents and provides their polarity index, viscosity, uv cutoff wavelength, solubility in water, and miscibility. The polarity index increases with polarity. Web this tool allows you interactively select solvents based upon the principal component analysis (pca) of the solvent's physical properties. Web because polar solvents stabilize the ground state more than the excited state, the change in energy increases with the polarity of the solvent. Web the polarity of a covalent bond can be judged by determining the difference of the electronegativities of the two atoms involved in the covalent bond, as summarized. However, novel and new substances or solvent combinations are.Solvent Miscibility and Polarity Chart
Polarity Chart Of Solvents
Polarity Of Solvents Chart
organic solvent polarity chart Bamil
Solvent Polarity Chart A Visual Reference of Charts Chart Master
Solvent Polarity Chart
Chemistry Solvent Characteristics CTG Clean
Solved Determine the solvent polarity index for each HPLC
Solved Determine the solvent polarity index for the
Polarity Chart Of Solvents
This Column Is Based On One Of The Most Widely Used Polar Phases, Carbowax™ 20M, And Is A Polar Column Suitable For Analyses Of Solvents, Fatty Acid.
Web 23 Rows Solvent Polarity;
Laboratory Techniques And Methods To.
It Also Includes Charts On Relative Solvent.
Related Post: