Water Vapour Pressure Chart
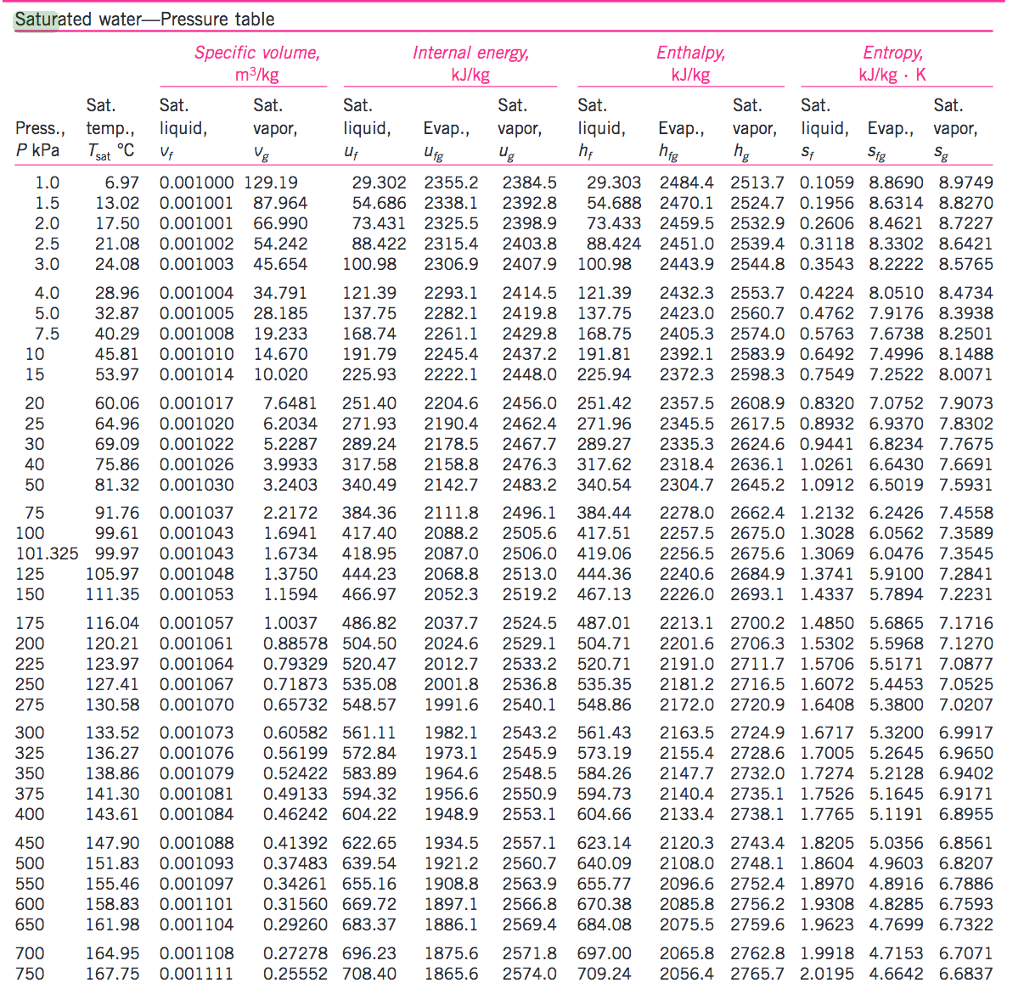
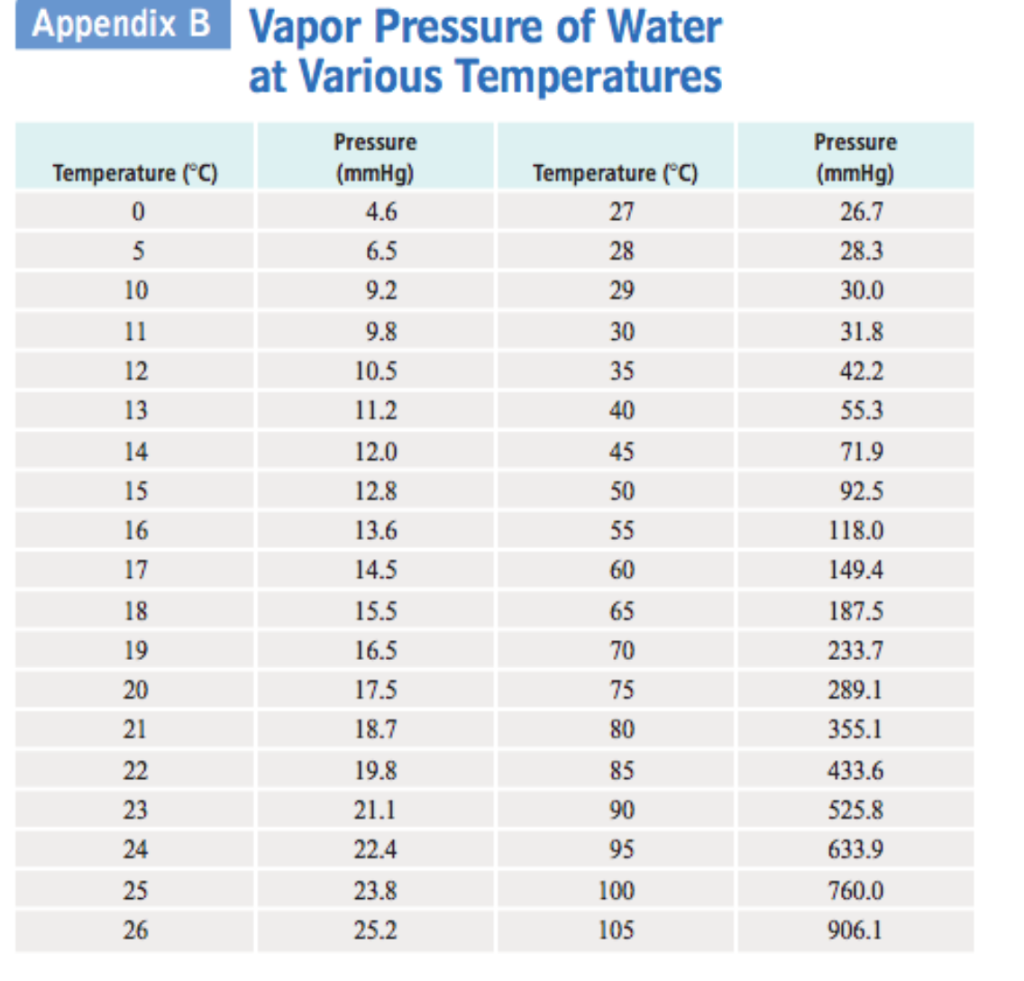
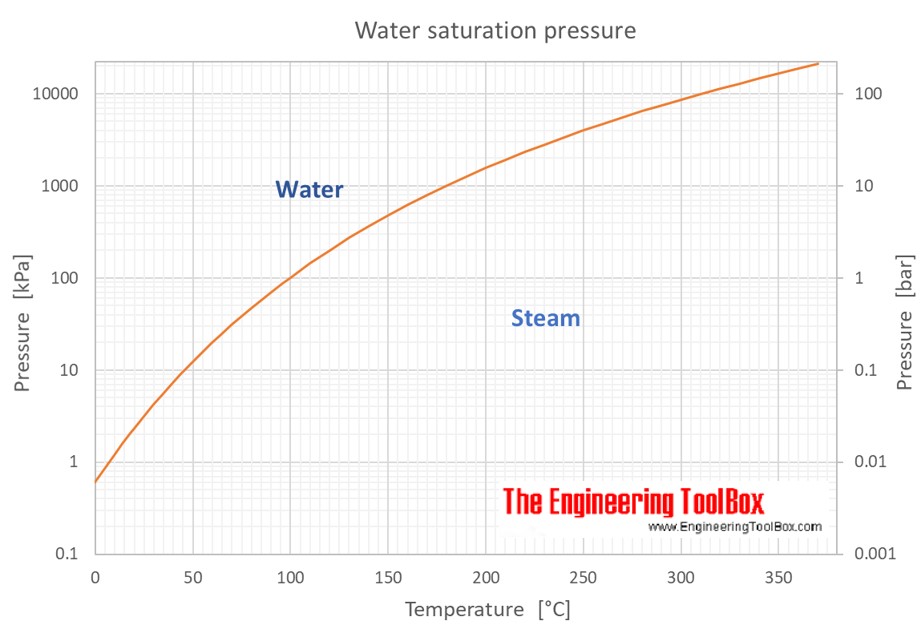
Water Vapour Pressure Chart - Web saturated vapor pressure, density for water. Web water vapour pressure table at different temperatures. Web vapor pressure of water (mmhg) source of data: Vapor pressure of water is shared under a not declared license and was authored, remixed, and/or curated by libretexts. Water at high pressure has a higher boiling point than when that water is at atmospheric pressure. Web with this vapor pressure of water calculator, you can find the vapor pressure at a particular temperature according to five different formulas. Web figure 2 2: Web vapor pressure of h 2 o at various temperatures (celsius) note that when water vapor pressure equals atmospheric pressure, then the water molecules are free to jump into the gas state. Vapor pressure is measured in the standard units of pressure. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and the water changes into a vapor. The saturation vapor pressure is the pressure at which water vapor is in thermodynamic equilibrium with its condensed state. Web vapor pressure of water (mmhg) at selected temperatures (°c) 0. Web vapor pressure of water (mmhg) source of data: Web explore a comprehensive table of water vapor pressure at different temperature values presented in both si (kpa) and us customary (psi) units. Vapor pressure is directly proportional to temperature). Below are some selected values of temperature and the saturated vapor pressures required to place the boiling point at those temperatures. Saturation (pressure) 11 p, mpa t, °c density, kg/m3 ρ l ρ v enthalpy, kj/kg h lv ∆h entropy, kj/(kg·k) s l s v ∆s volume, cm3/g v v 611.657 pa 0.01 999.79 0.004 855 0.00 2500.9 2500.9 0.000 00 9.1555 9.1555 1.000 21 205 991. Crc handbook of chemistry and physics, 84th edition (2004). At its boiling point (100 ° c), the vapor pressure of water is 658.0 torr (atmospheric pressure). Generally a substance's vapor pressure increases as temperature increases and decreases as temperature decreases (i.e. At its boiling point (100 ° c), the vapor pressure of water is 658.0 torr (atmospheric pressure). Vapor pressure is measured in the standard units of pressure. It is the pressure exerted by the saturated vapour in contact with the surface of the liquid at that temperature. Web this formula is illustrated in a phase diagram — a chart showing. It is the pressure exerted by the saturated vapour in contact with the surface of the liquid at that temperature. Web vapor pressure of water from 0 °c to 100 °c. Web vapor pressure of h 2 o at various temperatures (celsius) note that when water vapor pressure equals atmospheric pressure, then the water molecules are free to jump into. Pressure (degrees c) (mmhg) (degrees c) (mmhg) 0.0007 1.881 999.89 0.005 518 7.89 2504.3 2496.5 0.028 78 9.1058 9.0770 1.000 11 181 217. Web water vapour pressure table at different temperatures. Web vapor pressure of water from 0 °c to 100 °c. Web vapor pressure of h 2 o at various temperatures (celsius) note that when water vapor pressure equals. Generally a substance's vapor pressure increases as temperature increases and decreases as temperature decreases (i.e. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and the water changes into a vapor. 0.0007 1.881 999.89 0.005 518 7.89 2504.3 2496.5 0.028 78 9.1058 9.0770 1.000 11 181. Web the vapor pressure of water is the pressure exerted by molecules of water vapor in gaseous form (whether pure or in a mixture with other gases such as air). The pressure up cancels the pressure down and boiling begins. Pressure (degrees c) (mmhg) (degrees c) (mmhg) At its boiling point (100 ° c), the vapor pressure of water is. Web this formula is illustrated in a phase diagram — a chart showing how phases change and coexist at equilibrium at different pressures and temperatures. 0.0007 1.881 999.89 0.005 518 7.89 2504.3 2496.5 0.028 78 9.1058 9.0770 1.000 11 181 217. We look at the 68°f example specifically. Web water boiling temperature vs pressure in vacuum table chart. Web vapor. Web water boiling temperature vs pressure in vacuum table chart. Water at high pressure has a higher boiling point than when that water is at atmospheric pressure. Web explore a comprehensive table of water vapor pressure at different temperature values presented in both si (kpa) and us customary (psi) units. Web vapor pressure of water. Web figures and tables showing. Using this 1st calculator, you insert temperature in °f, and get the vapor pressure of water in terms of kpa, psi, mmhg, bar, atm, torr. Web the vapor pressure of water is the pressure exerted by molecules of water vapor in gaseous form (whether pure or in a mixture with other gases such as air). Web water vapour pressure table. Web for example, air at sea level, and saturated with water vapor at 20 °c, has partial pressures of about 2.3 kpa of water, 78 kpa of nitrogen, 21 kpa of oxygen and 0.9 kpa of argon, totaling 102.2 kpa, making the basis for standard atmospheric pressure. Web vapor pressure of water (mmhg) source of data: Vapor pressure of water. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and the water changes into a vapor. Web vapor pressure of water (mmhg) source of data: Web water boiling temperature vs pressure in vacuum table chart. 0.0007 1.881 999.89 0.005 518 7.89 2504.3 2496.5 0.028 78 9.1058. The boiling point of water is the temperature at which the vapor pressure of the liquid water equals the pressure surrounding the water and the water changes into a vapor. Web the vapor pressure of water at room temperature (25 ° c) is 23.8 mm hg, 0.0313 atm, or 23.8 torr, or 3.17 kpa. Web the vapor pressure of water is the pressure exerted by molecules of water vapor in gaseous form (whether pure or in a mixture with other gases such as air). At its freezing point (0 ° c), the vapor pressure of water is 4.6 torr. Vapor pressure is measured in the standard units of pressure. Web this formula is illustrated in a phase diagram — a chart showing how phases change and coexist at equilibrium at different pressures and temperatures. Web vapor pressure of water. Web water vapour pressure table at different temperatures. Web figure 2 2: Web explore a comprehensive table of water vapor pressure at different temperature values presented in both si (kpa) and us customary (psi) units. Vapor pressure is directly proportional to temperature). Vapour pressure is also called the vapour tension. Web vapor pressure of h 2 o at various temperatures (celsius) note that when water vapor pressure equals atmospheric pressure, then the water molecules are free to jump into the gas state. Saturation (pressure) 11 p, mpa t, °c density, kg/m3 ρ l ρ v enthalpy, kj/kg h lv ∆h entropy, kj/(kg·k) s l s v ∆s volume, cm3/g v v 611.657 pa 0.01 999.79 0.004 855 0.00 2500.9 2500.9 0.000 00 9.1555 9.1555 1.000 21 205 991. At its boiling point (100 ° c), the vapor pressure of water is 658.0 torr (atmospheric pressure). Water at high pressure has a higher boiling point than when that water is at atmospheric pressure.Water Vapour Pressure Chart Bar
How do you find vapor pressure of water at given temperature? Socratic
Water Vapour Pressure Chart Bar
Vapour Pressure Of Water Chart
Vapor Pressure Chart For Water
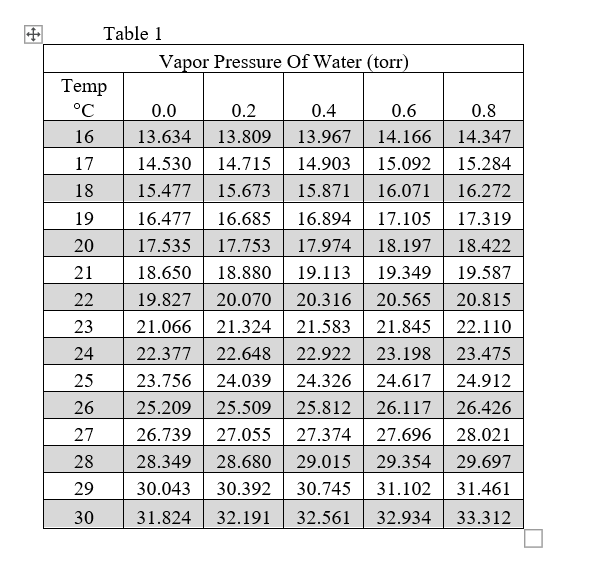
[PDF] Vapor Pressure Formulation for Water in Range 0 to 100 °C. A
Vapour Pressure Of Water Chart
Vapor Pressure Chart For Water
Conservation physics Fundamental microclimate concepts
Vapor Pressure Chart For Water
Generally A Substance's Vapor Pressure Increases As Temperature Increases And Decreases As Temperature Decreases (I.e.
The Pressure Up Cancels The Pressure Down And Boiling Begins.
Crc Handbook Of Chemistry And Physics, 84Th Edition (2004).
It Is The Pressure Exerted By The Saturated Vapour In Contact With The Surface Of The Liquid At That Temperature.
Related Post:

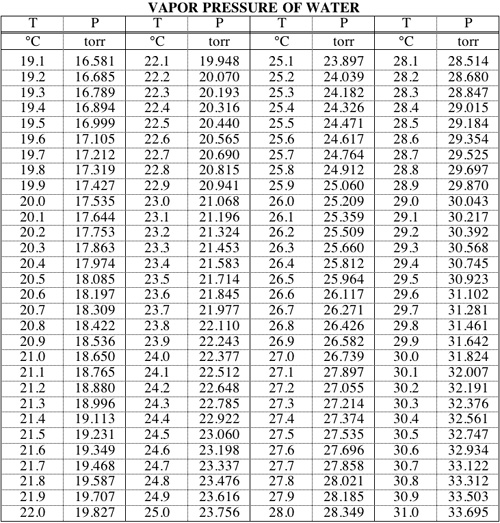

![[PDF] Vapor Pressure Formulation for Water in Range 0 to 100 °C. A](https://d3i71xaburhd42.cloudfront.net/471011a1b864be3f78720f4d8ba4f21385acd117/8-Table7-1.png)